What happens when excess CO2 is passed through lime water is a common chemistry question with an interesting answer. At first, lime water turns milky because carbon dioxide reacts with calcium hydroxide and forms calcium carbonate. This white solid makes the solution look cloudy. When more carbon dioxide is passed through the same lime water, the milky color slowly disappears. This happens because calcium carbonate changes into calcium bicarbonate, which dissolves in water. This reaction is commonly used in chemistry labs and school experiments to test the presence of carbon dioxide gas.
This process is a great example of how chemical reactions can change when the amount of gas increases. In the first step, a white precipitate is formed, while in the second step, that solid disappears into a clear solution. Students often perform this experiment to understand precipitation and soluble compounds in an easy way. It also helps explain why lime water is an important indicator for detecting carbon dioxide in science experiments.
What Happens When Excess CO2 Is Passed Through Lime Water?
What happens when excess CO2 is passed through lime water can be explained in two simple steps. First, the clear lime water turns milky. After more carbon dioxide is added, the milky color disappears and the solution becomes clear again.
First Change: Lime Water Turns Milky
At the start, carbon dioxide reacts with lime water, which contains calcium hydroxide. This reaction forms calcium carbonate, a white solid that makes the liquid look milky.
Ca(OH)_2 + CO_2 \rightarrow CaCO_3 + H_2O
Second Change: Milkiness Disappears
When more CO2 keeps passing through the solution, the calcium carbonate reacts again and changes into calcium bicarbonate. This new compound dissolves in water, so the solution becomes clear.
CaCO_3 + CO_2 + H_2O \rightarrow Ca(HCO_3)_2
Final Answer in One Line
So, what happens when excess CO2 is passed through lime water is that it first turns milky and then becomes clear when more carbon dioxide is passed through it.
Why What Happens When Excess CO2 Is Passed Through Lime Water Causes Milky Color First
Many students ask why the solution becomes cloudy before turning clear. When learning what happens when excess CO2 is passed through lime water, this first milky change is the most important step.
Carbon Dioxide Reacts with Lime Water
Lime water is a clear solution of calcium hydroxide. When carbon dioxide enters the liquid, it reacts quickly and forms calcium carbonate.
Ca(OH)_2 + CO_2 \rightarrow CaCO_3 + H_2O
White Solid Makes the Water Milky
Calcium carbonate does not dissolve easily in water. It appears as tiny white particles floating in the solution. These particles make the lime water look milky or cloudy.
Why This Step Matters
This milky color shows that carbon dioxide is present. That is why schools and laboratories use this reaction as a simple gas test.
Simple Answer
So, what happens when excess CO2 is passed through lime water causes a milky color first because insoluble calcium carbonate is formed.
What Happens When Excess CO2 Is Passed Through Lime Water After More Gas Is Added
After the first milky stage, the reaction changes again. To fully understand what happens when excess CO2 is passed through lime water, you need to know what occurs when extra gas continues to bubble through the solution.
Milky Solution Starts to Clear
At first, calcium carbonate forms and makes the liquid cloudy. When more carbon dioxide is added, this white solid reacts with water and extra CO2.
CaCO_3 + CO_2 + H_2O \rightarrow Ca(HCO_3)_2
A Soluble Compound Is Formed
The new product is calcium bicarbonate. It dissolves in water, so the white particles disappear and the solution becomes clear again.
Why This Happens
Extra carbon dioxide changes the insoluble calcium carbonate into a soluble substance. Because it dissolves, the cloudy look is removed.
Simple Answer
So, what happens when excess CO2 is passed through lime water after more gas is added is that the milkiness disappears and the solution turns clear.
Chemical Reaction Behind What Happens When Excess CO2 Is Passed Through Lime Water
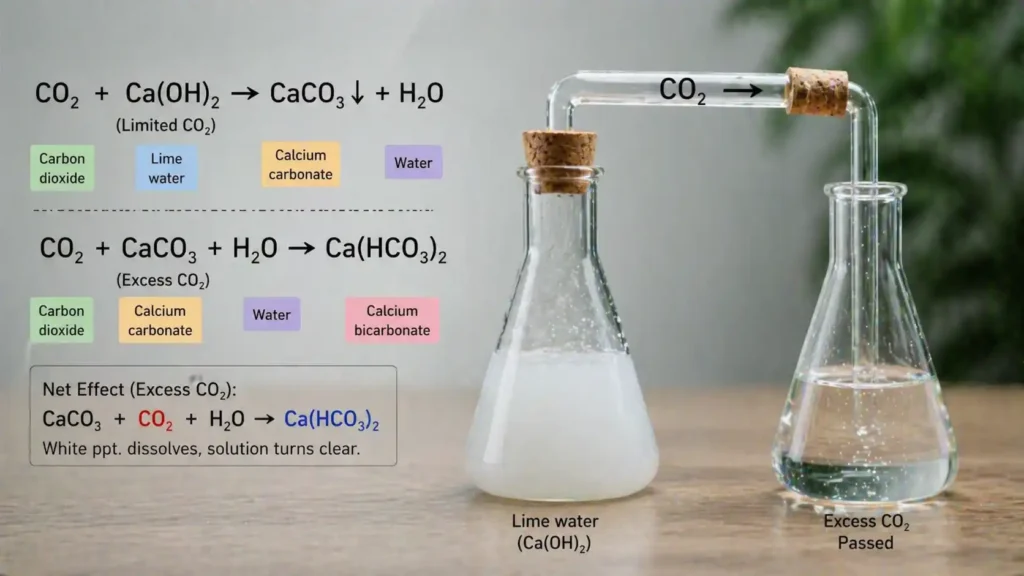
To understand what happens when excess CO2 is passed through lime water, it is helpful to know the chemical reactions involved. This process happens in two clear stages.
First Reaction: Formation of Calcium Carbonate
When carbon dioxide is passed through lime water, it reacts with calcium hydroxide. This forms calcium carbonate, which makes the solution milky.
Ca(OH)_2 + CO_2 \rightarrow CaCO_3 + H_2O
Second Reaction: Formation of Calcium Bicarbonate
When excess carbon dioxide is added, the calcium carbonate reacts again. It changes into calcium bicarbonate, which dissolves in water.
CaCO_3 + CO_2 + H_2O \rightarrow Ca(HCO_3)_2
Why Two Reactions Matter
The first reaction causes milkiness, while the second reaction removes it. This is why the solution first turns cloudy and then becomes clear.
What Happens When Excess CO2 Is Passed Through Lime Water in School Experiments
This reaction is often shown in science classes because it is simple and easy to observe. Students can clearly see what happens when excess CO2 is passed through lime water through the color changes in the solution.
How the Experiment Is Done
Teachers usually pass carbon dioxide gas through a test tube containing clear lime water. The gas may come from breathing out through a straw or from a chemical reaction in the lab.
What Students Observe First
At the start, the clear lime water turns milky. This happens because calcium carbonate is formed.
Ca(OH)_2 + CO_2 \rightarrow CaCO_3 + H_2O
What Happens with Excess CO2
If carbon dioxide keeps passing through the liquid, the milkiness disappears. The solution becomes clear again because calcium bicarbonate is formed.
CaCO_3 + CO_2 + H_2O \rightarrow Ca(HCO_3)_2
Why This Experiment Is Popular
- Easy to perform
- Clear visual change
- Helps students understand gas reactions
- Used to test carbon dioxide presence
Factors That Affect What Happens When Excess CO2 Is Passed Through Lime Water
Several conditions can change the speed and result of the reaction. To understand what happens when excess CO2 is passed through lime water, it is useful to know these important factors.
Amount of Carbon Dioxide
If only a small amount of CO2 is added, lime water turns milky. If excess CO2 is passed, the milkiness disappears and the solution becomes clear.
Strength of Lime Water
Fresh and properly prepared lime water gives better results. Weak or old lime water may show slower or lighter changes.
Speed of Gas Flow
If carbon dioxide enters slowly, the reaction is easier to observe. Very fast bubbling may make the changes harder to notice.
Temperature of the Solution
Temperature can affect how quickly the reaction happens. Normal room temperature is best for clear results in school experiments.
Clean Equipment
Dirty test tubes or impure chemicals can affect the final appearance of the solution.
Simple Answer
So, the main factors that affect what happens when excess CO2 is passed through lime water are CO2 amount, lime water strength, gas flow speed, temperature, and clean equipment.
Real-Life Uses of What Happens When Excess CO2 Is Passed Through Lime Water Test
The lime water reaction is not only for classrooms. It also helps in practical situations. Knowing what happens when excess CO2 is passed through lime water shows why this test is useful in daily life and science.
Detecting Carbon Dioxide Gas
Lime water is commonly used to check if a gas contains carbon dioxide. If the solution turns milky, CO2 is present.
Breathing and Respiration Experiments
Students blow air through lime water to show that exhaled air contains carbon dioxide. This is a common science demonstration.
Industrial and Laboratory Use
Some labs use lime water tests during experiments where carbon dioxide may be produced.
Environmental Learning
This reaction helps explain how carbon dioxide behaves in nature and water systems.
Why It Is Useful
- Simple and low cost
- Easy to observe
- Fast results
- Good for teaching chemistry basics
Simple Answer
So, real-life uses of what happens when excess CO2 is passed through lime water include gas testing, respiration experiments, laboratory work, and science education.
Common Mistakes About What Happens When Excess CO2 Is Passed Through Lime Water
Many students get confused about this reaction. Learning the common mistakes helps you better understand what happens when excess CO2 is passed through lime water.
Thinking the Solution Stays Milky Forever
Some people believe lime water remains cloudy all the time. This is incorrect. With excess CO2, the milkiness disappears and the solution becomes clear again.
Mixing Up the Products
A common mistake is thinking the same product forms in every step. First, calcium carbonate is formed. Later, calcium bicarbonate is produced.
Forgetting the Two Stages
This reaction happens in two parts:
- First stage: milky solution
- Second stage: clear solution
Using Old Lime Water
Old or weak lime water may not give clear results. Fresh lime water is best for proper observation.
Ignoring Excess CO2
If only a small amount of carbon dioxide is used, you will not see the final clear stage.
Simple Answer
So, common mistakes about what happens when excess CO2 is passed through lime water include forgetting the two-step reaction, using weak lime water, and expecting the solution to stay milky.
FAQs
1. What happens when excess CO2 is passed through lime water first?
At first, lime water turns milky because calcium carbonate is formed during the reaction.
2. Why does lime water turn milky?
Lime water turns milky because white calcium carbonate particles are produced.
3. Why does milkiness disappear in excess CO2?
The milkiness disappears because calcium carbonate changes into soluble calcium bicarbonate.
4. What is the formula of lime water?
Lime water is a solution of calcium hydroxide.
Ca(OH)_2
5. Where is this reaction used?
This reaction is used in school experiments, chemistry labs, and for testing carbon dioxide gas.
Conclusion
Now you know what happens when excess CO2 is passed through lime water. First, the clear lime water turns milky because calcium carbonate is formed. When more carbon dioxide is added, the milkiness disappears because calcium bicarbonate is produced and dissolves in water. This simple two-step reaction is important in chemistry and is widely used in school experiments to test carbon dioxide gas. It is easy to remember: first cloudy, then clear.